Immune checkpoint inhibitors (ICIs) have emerged as revolutionaries in the ever-evolving field of cancer therapy. By harnessing the body’s own immune system to fight cancer, they have marked a significant advancement in immunotherapy. While traditional therapies kill cancer cells directly, these drugs work by inducing immune cells to detect and destroy tumors with remarkable precision. This innovation is reshaping how advanced and metastatic cancers are treated, offering hope where there was little before.
The immune system’s delicate balance and cancer’s stealth tactics
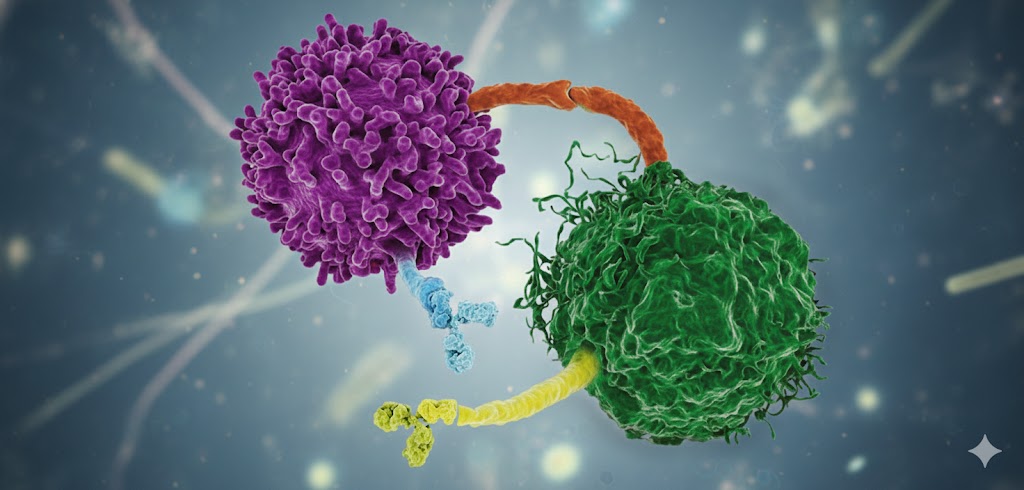
Our immune system, particularly T-cells, play an important role in identifying and eliminating abnormal cells, including cancerous ones1. These T-cells are trained to recognize and destroy threats. However, the immune system must also avoid attacking the body’s own healthy tissues—a balance strictly maintained by immune checkpoint proteins.
These checkpoint proteins, such as CTLA-4 and PD-1, act as natural “off switches” for T-cells2. They mainly prevent unwarranted immune reactions and autoimmune disease by turning off immune responses once the threat has cleared. Thus, they serve as crucial brakes on immune activity, maintaining harmony within the body, preventing unnecessary triggers.
Cancer cells have evolved to turn this carefully orchestrated system to their advantage. They bind to checkpoint receptors such as PD-1 on T-cells by expressing proteins like PD-L1, sending inhibitory signals that effectively silence immune attacks. This facilitates tumors to create an immunosuppressive environment, escaping immune surveillance and growing unchecked3. What ensues is a stealthy cancer that evades one of the body’s most powerful defense mechanisms.
How do immune checkpoint inhibitors work?
Immune checkpoint inhibitors are specially engineered therapeutic monoclonal antibodies, designed to block these key checkpoint proteins. They do so by releasing the brake on T-cells and reviving the ability of the immune system to detect and kill tumor cells. These antibodies represent one aspect of the expanding immunotherapeutic machinery, along with treatments like CAR T-cell therapy.
There are two major classes of ICIs:
CTLA-4 inhibitors
They work by blocking the CTLA-4 protein, found on T-cells. This protein’s primary function is to regulate the early activation of T-cells within the lymph nodes. By inhibiting CTLA-4, these drugs increase the number of active T-cells, making them more available to recognize and attack cancer cells. An example of this type of drug is ipilimumab, which blocks the CTLA-4 protein on T-cells4. CTLA-4 primarily regulates the early activation of T-cells in lymph nodes. By inhibiting CTLA-4, these drugs increase the pool of active T-cells ready to recognize cancer.
PD-1/PD-L1 inhibitors
They work by blocking the PD-1 receptor on T-cells, such as, with pembrolizumab and nivolumab, or its ligand PD-L1 on cancer cells as seen with atezolizumab and durvalumab5-8. This prevents tumors from switching off T-cells that have already infiltrated the tumor, effectively reactivating immune attack within the tumor microenvironment.
Together, these inhibitors restore the immune system’s ability to eliminate cancer, transforming the patient’s body into a powerful anti-cancer weapon of sorts.
Transformative clinical successes across multiple cancers
Initially approved for advanced melanoma, ICIs have rapidly expanded their reach to numerous cancer types. Their impact on patient outcomes has been striking:
- Once a cause for concern, advanced melanoma now sees significantly improved survival with ICIs9.
- ICIs have become a standard frontline therapy for Non-Small Cell Lung Cancer (NSCLC)10.
- Often used in combination therapies, ICIs have delivered meaningful benefit in the treatment of renal cell carcinoma11.
- ICIs provide survival benefits in recurrent or metastatic Head and neck squamous cell carcinoma (HNSCC) by reactivating the immune system to target cancer cells, though response rates remain modest12.
- ICIs have revolutionized treatment for both advanced and metastatic urothelial carcinoma, offering clinically significant as well as FDA-approved for patients unresponsive to standard therapies13. They do this particularly by targeting the PD-1/PD-L1 and CTLA-4 pathways.
- Microsatellite instability-high or mismatch repair deficient cancers, ICIs have a breakthrough tumor-agnostic approval, meaning any solid tumor with these biomarkers, regardless of origin—can be treated14.
- Hodgkin lymphoma, liver cancer (hepatocellular carcinoma), esophageal and gastric cancers, among otheOne of the most remarkable features of ICIs is their ability to induce enduring remissions15. Some patients can keep their cancer under control for years even after stopping treatment, highlighting the importance of “immune memory” that guards against any relapse.
Challenges and immune-related side effects
Despite the promise they offer, ICIs come with their set of drawbacks. These drugs are capable vof launching a full-blown attack on perfectly healthy tissues, leading to immune-related adverse events (irAEs), which can affect nearly any organ16:
- Skin reactions, including rashes
- Gastrointestinal inflammation causing colitis, diarrhea, and pain
- Endocrine disorders such as thyroid dysfunction, adrenal insufficiency, and pituitary inflammation
- Liver inflammation (hepatitis)
- Lung inflammation (pneumonitis)
- Musculoskeletal issues like arthritis
- Neurological complications, though rarer, can be serious (for example, neuropathy, myasthenia gravis)
When it comes to managing irAEs effectively, it is important to recognize and intervene early on, often with immunosuppressive therapies such as corticosteroids17. In some cases, ICIs must be discontinued permanently to prevent further harm, if deemed fit.
Indeed, not all patients benefit from ICIs. Primary resistance and acquired resistance, mainly, are the factors that pose serious challenges18. Biomarkers such as PD-L1 expression, tumor mutational burden, and MSI status do help guide treatment but are imperfect predictors19. The risk of pseudoprogression—where tumors appear to grow on scans before shrinking, further complicates assessment20. The high cost of ICIs also poses as a formidable barrier to their routine usage21.
Future directions
Research into ICIs continues at a remarkable pace, focused on enhancing efficacy, overcoming resistance, and reducing toxicity. Combination therapies are a key strategy, pairing ICIs with22:
- Other checkpoint inhibitors, for example, combining CTLA-4 and PD-1 inhibitors to boost response rates in cancers like melanoma and kidney cancer23
- Chemotherapy, which can make tumors more visible to the immune system by causing immunogenic cell death24
- Radiation therapy, which releases tumor antigens and creates inflammation that sensitizes tumors to ICIs25
- Targeted therapies focused on molecular abnormalities within cancer cells
- Additional immunotherapies such as cancer vaccines or oncolytic viruses that stimulate immune responses26, 27
- Emerging technologies like theranostics and nanomedicine enable simultaneous diagnosis and treatment, enhancing delivery and monitoring of immunotherapies28, 29.
- Disease modeling using organoids—3D cultures mimicking patient tumors, allows personalized testing of immunotherapeutic responses before clinical application30.
- Additionally, multi-omics approaches—integrating genomics, proteomics, and metabolomics, along with artificial intelligence are becoming pivotal to better understand tumor heterogeneity, identify biomarkers, and tailor treatments, advancing the promise of precision medicine31, 32.
Beyond PD-1 and CTLA-4, new checkpoints like LAG-3, TIM-3, and TIGIT are being explored as drug targets, along with stimulatory pathways (OX40, CD40) aimed at further activating immune cells33-36.
Efforts to develop more reliable biomarkers are intensifying, including studies of the gut microbiome and blood-based markers to personalize therapy and anticipate side effects37-39. Understanding why some tumors resist ICIs is vital for new strategies to boost effectiveness.
Importantly, there is growing interest for using ICIs in the early stages of a disease—in adjuvant (post-surgery) and neoadjuvant (pre-surgery) settings, to improve long-term outcomes and prevent recurrence40.
Conclusion
ICIs have revolutionized cancer treatment by empowering the immune system to fight tumors, offering new hope and improved survival for patients. While challenges still remain, these therapies represent a new era of personalized, immune-based cancer care.
Meta title: Immune Checkpoint Inhibitors: Unlocking Cancer Defense
Meta description: Understanding how immune checkpoint inhibitors unleash T-cells to destroy tumors, transforming cancer treatment with new, durable responses.